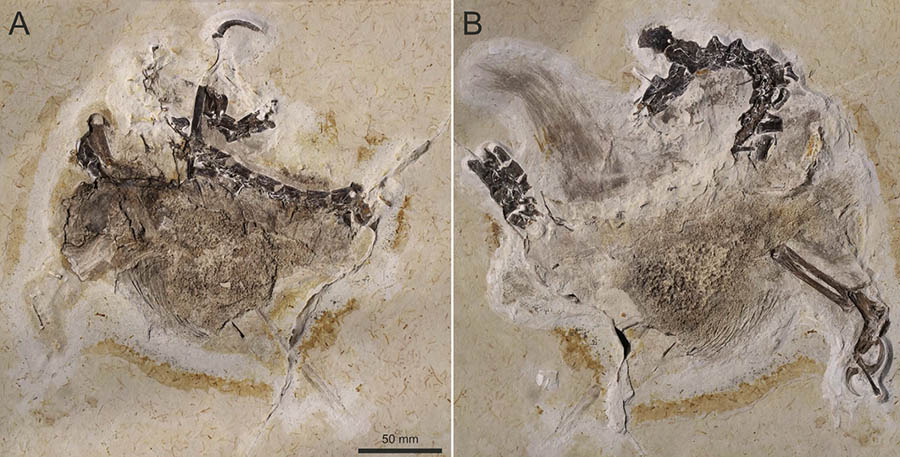
'Jurassic World Aftermath' Lets You Play VR Hide-and-Seek With a Cartoon Velociraptor

Ever catch yourself watching that fraught kitchen scene with the kids from the first Jurassic Park film and wondering how well you’d handle a face-off against a cunning velociraptor? If so, then the new Oculus Studios exclusive Jurassic World Aftermath is your chance to test those nerves of steel.
“It’s about evasion,” says Brian Gomez, executive producer for Universal Games and Digital Platforms, of Aftermath’s stealth-and-puzzle-based gameplay. “It’s about really learning the velociraptor’s [thinking] … being able to read its body language. You’re going to spend a lot of time sneaking around the facility, hiding under desks, hiding in lockers, hiding in ventilation shafts, and observing.”
Gomez isn’t underselling the virtual reality experience. Aftermath, which launches this Thursday, Dec. 17 for $25 on the Quest platform, is primarily a game of cat and mouse that takes place on the fictional-but-familiar-to-Jurassic-fans setting of Isla Nublar. The game, developed by indie studio Coatsink, puts players in the shoes of Sam — a security expert tasked with infiltrating the ruins of notorious genetics company InGen’s dinosaur-ridden facility, in order to pillage trade secrets from its servers. The premise sounds straightforward enough except for the fact that you’re being hunted the entire time by wily velociraptors who can hear, and remember, your every noisy move.
“There were countless hours spent trying to balance the velociraptor because she was just so damn good,” Gomez says of the enemy dino’s artificial intelligence. “AI really [progressed] to the point where it was very few of us that could actually survive her. And I think we finally got to the point where, when you first start, you're probably going to die a lot. You're going to die a lot until you start learning her behavior and … reading her body language and audio cues.”
Aftermath’s sound design is absolutely crucial to successfully navigating the experience, so it’s best to play with headphones. Dr. Mia Everett, your offscreen partner (voiced by Laura Bailey of The Last of Us: Part II fame), will guide you through the experience, providing you with audio-based story updates and mission objectives. To progress, you’ll often need to pause and listen carefully to nearby sounds like approaching footsteps, clanging objects being knocked over, or the disquieting chirps of the chicken-sized compsognathus before blindly barreling ahead. Even something as innocuous as the rumbling growl of a nearby apatosaurus is enough to send players into a mild panic.

Oh, and if you run too much, the noise you make will trigger the velociraptor’s curiosity and you’ll soon find yourself set on a red-screened course for imminent death. Instead, it’s best to play Aftermath slowly and quietly, relying on your wrist-worn compass for navigation and taking advantage of environmental triggers like an alarm or radio to distract the velociraptor and throw her off your trail while you solve puzzles.
“You're essentially ringing the dinner bell for a velociraptor to go to a feeding station or something and go to an area of the lab,” Gomez says. “And that'll buy you 10 seconds to run to a new hiding space or do the thing you've got to do and get the hell out.”
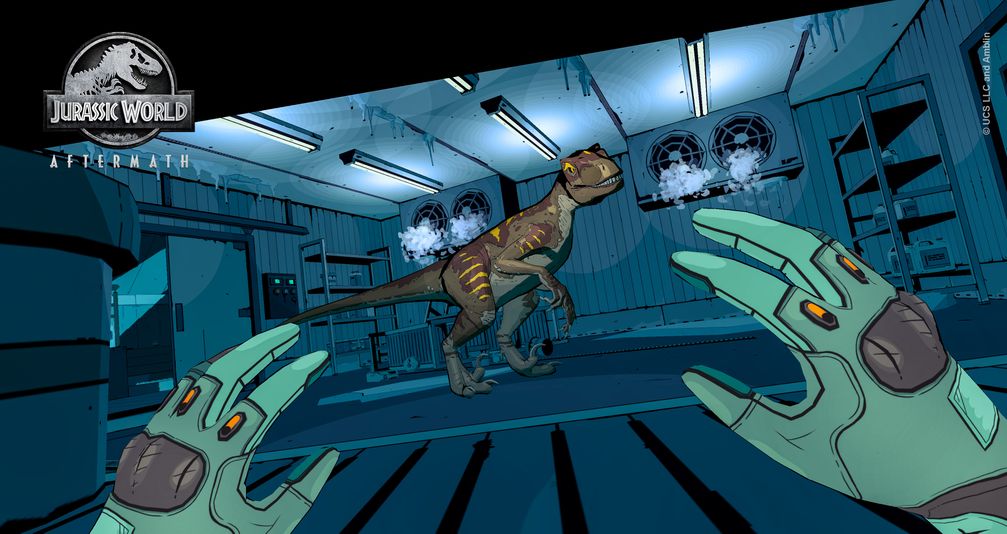
Rest assured Aftermath isn’t straight-up VR horror, so players of varying degrees of mental fortitude will be able to stomach the game’s panic-inducing scenarios. Personally, I worried it would prove too intense for my tastes. After all, I still haven’t been able to beat Half Life: Alyx’s “Jeff” level and I also noped out of Alien: Isolation, both of which employ gameplay elements similar to Aftermath. But after a tense initial hour of play during which I remained in a crouch pose and very, very, very cautiously moved throughout the facility while occasionally turning noisy cranks so painfully slowly that it seemed five minutes had passed, my anxiety lessened. Aftermath isn’t a cheap game rife with jump scares akin to Five Nights at Freddy’s. If you make too much noise and alert the velociraptors, well, then that’s on you.
Coatsink worked closely with Gomez’s team at Universal to ensure a delicate balance to Aftermath’s fear-based gameplay so that it would be welcoming to all fans of the Jurassic franchise.
“We just tried to strike a similar tone to the films because the films often move seamlessly from those moments of terror into those moments of wonder,” he says.
“There's times where you've just spent the last 20 minutes playing hide-and-seek with a velociraptor and it's sort of super intense. And then you'll get to an area of the lab where you look out the window and there's a flock of pterodactyls flying by.”
Easing that tension somewhat is the game’s distinct, comic-book-like art style. The simple, flat-shaded look of the environments and dinosaurs, with their thick-inked outlines, helps to dial down the terror, making it easier for nervous players (like myself) to progress. And it makes sense: A one-on-one encounter with a cartoon velociraptor is much more tolerable for audiences than one with glistening teeth, flaring nostrils, and a feathery coat. It’s an intentional design shift away from the Jurassic franchise’s bread-and-butter aesthetic of realistic CGI dinosaurs.
“We wanted it to still be scary and affecting and immersive, even if it wasn't filmic — like photo-realism. But we also didn't want it to be so intimidating and so scary … [for] a huge portion of the Jurassic audience that doesn't want a totally visceral R-rated horror experience,” Gomez says.
But reducing the terror level isn’t the sole reason for Aftermath’s signature look, although it is a nice bonus. Gomez says it mostly has to do with the game’s art, AI, and sprawling environmental design fitting within the technological constraints of the mobile chipsets powering the Quest and Quest 2 headsets. This, he says, allowed the team “to create the Raptor behavior and the size of the facility.”
Players looking for a meaty, multi-hour stealth experience may be disappointed by Aftermath’s length. The game can be completed in about three hours’ time, but Gomez cautions that’s only for those who follow the “golden path,” making little to no mistakes and ignoring optional story content. I’ve logged about two hours with Aftermath so far and I’m only just past the first main objective. But, again, I’m playing very carefully because I don’t do well with horror, so your total playtime will obviously vary. If you find yourself wanting more after a full playthrough, hold tight — Coatsink plans to release a Part 2 DLC sometime in 2021 which, Gomez teases, will ramp up “the danger and the intensity.”

As for play style options, Aftermath offers the ability to toggle between standing or seated mode, as well as three comfort presets: comfortable, moderate, and intense. There’s also a custom option which lets you toggle your preferred settings for movement (sorry, there’s no teleport!), turning, and vignette strength. I’ve been playing on the moderate setting which adds a mild vignette to your peripheral vision while you move about the environment, and have had no issues whatsoever.
Oh, and for those looking for familiar faces from the Jurassic film franchise sprinkled throughout the game, I’m sorry to say you won’t find any in Aftermath. But you will hear them. That’s right, Jeff Goldblum and B.D. Wong have recorded original dialogue for the game, reprising their roles as Dr. Ian Malcolm and Dr. Henry Wu, respectively.
No word on a Laura Dern easter egg, though. (A boy can dream.)
Source: https://mashable.com/